New COVID-19 drug therapies are on the horizon. But will they matter?
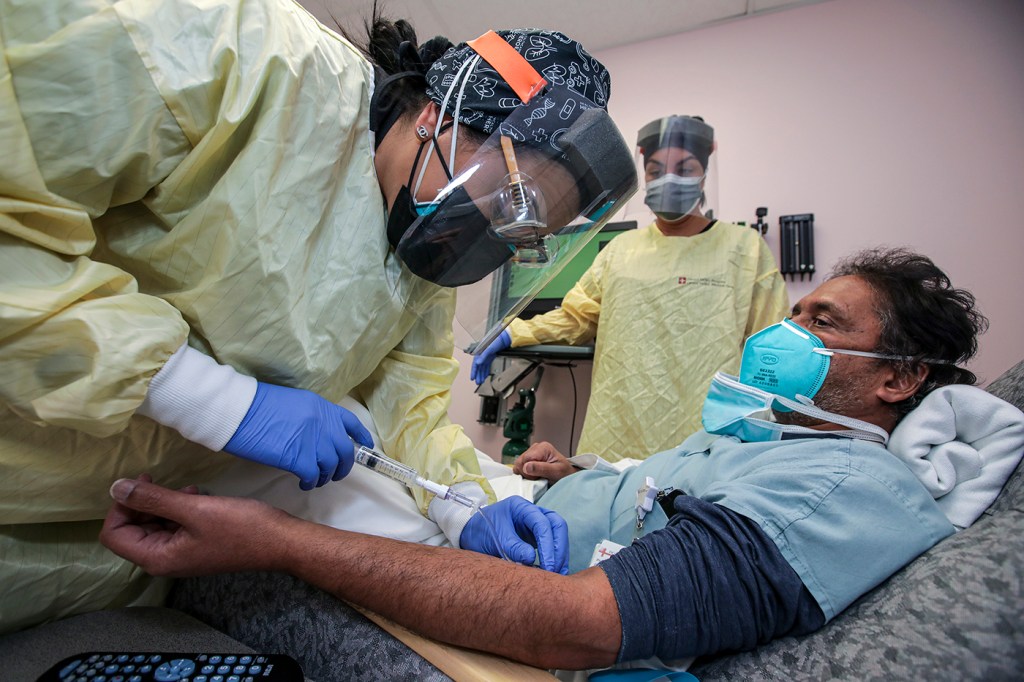
With new COVID-19 cases remaining at high levels across the U.S., new drug therapies that may help people who contract the respiratory disease evade serious illness are starting to garner more attention.
The World Health Organization recently endorsed an antibody drug cocktail made by Regeneron Pharmaceuticals, a combination drug that has been used widely to bolster sick patients by infusing them with antibodies designed to combat COVID. And a new antiviral pill that could be taken daily, called molnupiravir, is being studied as a way of halting COVID illness early in its course. Other therapies, such as the potential to extract COVID “nanobodies” from llamas, are also being explored.

Mansoor Amiji, Distinguished Professor and Chair of the Department of Pharmaceutical Sciences and Brandon Dionne, Assistant Clinical Professor Department of Pharmacy and Health Sciences, pose for a portrait. Photos by Matthew Modoono/Northeastern University
While some existing treatments for COVID have helped to shore up hospital resources by preventing severe illness in many patients, they are far from a silver bullet, and face numerous production and efficacy challenges, say several Northeastern experts.
“That’s the challenge with respiratory viruses—it’s difficult to develop a good antiviral for them,” says Brandon Dionne, associate clinical professor of pharmacy and health systems sciences at Northeastern. “And there’s not as much incentive in the market” to pursue these therapies.
Treatments for COVID are classified into several categories: antibody therapies, which use lab-produced antibodies to help stop viral replication; antivirals, which specifically target and kill viral particles; and corticosteroids, which can help reduce the body’s inflammatory response to the infection.
Antibody treatments like Regeneron’s, which combines casirivimab and imdevimab into a “monoclonal antibody cocktail” have been shown to be effective in reducing COVID-19 hospitalization, though it must be used early in the course of illness, Dionne says. The drug cocktail was also shown to have helped resolve COVID symptoms and reduced the viral load in patients, according to data published Wednesday in the New England Journal of Medicine.
And some Massachusetts hospitals have touted the therapy, saying it’s helped to save lives during the summer surge. But states are starting to limit its use only to high-risk patients, particularly the unvaccinated, amid an ongoing supply shortage. About 70 percent of the national supply of the antibody treatments have gone to states hardest hit in recent months, including Tennessee, Alabama, Mississippi, Florida, Georgia, Louisiana, and Texas.
Additionally, the timing of treatment is crucially linked to outcomes, in both the use of monoclonal antibody therapies and in antivirals. There is also concern about how effective these treatments will fare against an ever-evolving virus, says Mansoor Amiji, university distinguished professor of pharmaceutical sciences and chemical engineering at Northeastern.
As with the vaccines, the drug therapies “have some affinities to the viral strain against which they were originally tested,” Amiji says.
“The virus is evolving,” Amiji adds. “It’s trying to resist, both on the vaccine side and the drug side. So there is potentially a concern that both the antibodies and these [antiviral] drugs would not be so effective in the long run.”
Amiji says research and development into new drug therapies is happening, but only at the pace of investment, which is lagging behind because of the focus on vaccines. Amiji hopes that starts to change, noting that new treatments may also help to mitigate or prevent symptoms in so-called “long haulers,” a condition marked by prolonged COVID symptoms that could impact as many as one-third of those who become infected.
“The financial incentives are not there,” he says. “New research is needed to develop these novel antivirals.”
The only Food and Drug Administration approved antiviral for COVID, and the only fully approved treatment, is Remdesivir, which has also been shown to reduce hospitalization by up to 87 percent in high risk patients if administered early. But there is no evidence to suggest Remdesivir reduces COVID mortality, according to the World Health Organization.
Remdesivir is not a new drug. Originally developed to treat Hepatitis C, Amiji says the treatment had been shelved for years before it was repurposed to fight COVID. The drug is not intended for widespread use, and can only be given intravenously, further limiting who can use it.
Monoclonal antibody therapies are also administered intravenously. Both treatment forms appear to have little to no effect on patients during the latter stages of disease progression, experts say.
That’s when various steroid drugs would be used, which Dionne says do have a mortality benefit.
The prospect of the pill-form of molnupiravir is a promising development in that it could be used outside hospital settings, increasing treatment options for many people. But Dionne says all drug therapies have so far proved to be only “modestly effective” compared to the overwhelming efficacy of the vaccines.
“They do play a role,” Dionne says, “but vaccination is by the far the most important thing here.”
For media inquiries, please contact media@northeastern.edu.






