This superbug dodges antibiotics. He’s finding a way to kill it.

It’s a daily occurrence. A patient goes into a hospital for, say, a broken bone and then is infected by a so-called superbug that spreads easily around a hospital.
About one in every 31 hospital patients has at least one infection that they contracted in a healthcare facility on any given day, according to the U.S. Centers for Disease Control and Prevention. And many of those pathogens are resistant to antibiotic treatment, which means that the infection can progress to cause serious issues such as pneumonia or sepsis.
So how, then, can doctors treat patients who become infected by these bacteria?
That’s a question that gnawed at Edward Geisinger when he was doing infectious disease rotations during medical school. He saw many patients in critical care wards that had been infected by a bacterium called Acinetobacter baumannii, which is highly resistant to antibiotics.

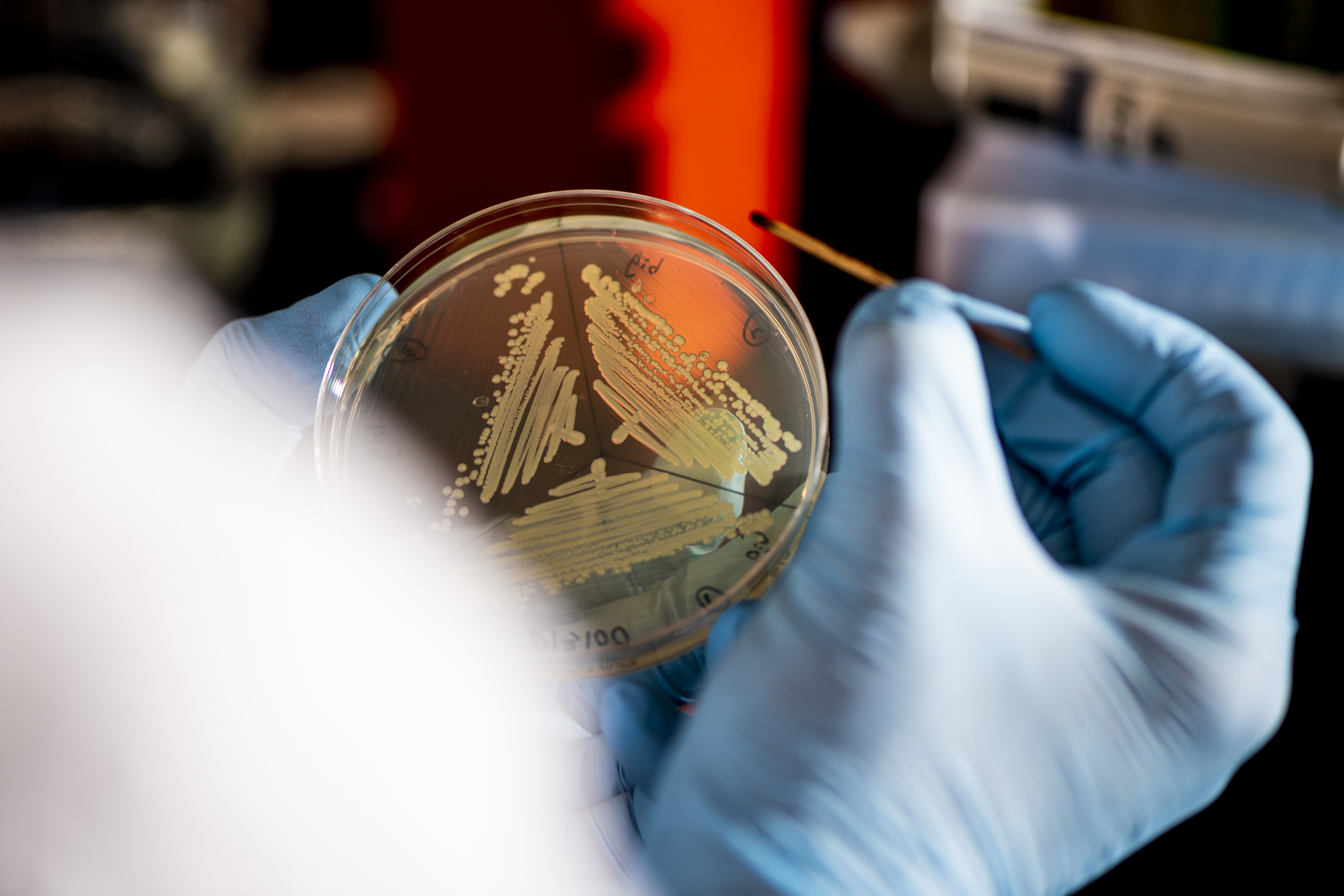
“I knew it was becoming a problem, but was an understudied microbe at the time,” says Geisinger, now assistant professor of biology at Northeastern. So after he finished his medical and doctoral coursework, he began to study the bacterium in depth.
“In the clinic, it causes pneumonia associated with people on ventilators, and bloodstream infections that can progress into sepsis,” he says. “Usually it infects the sickest people in the hospital. They’re really ill, so their whole immune system is sort of struggling. And they’re also hooked up to various devices, so organisms have portals of entry.”
Little was known about A. baumannii, beyond the havoc it could wreak on patients, so Geisinger’s research initially focused on simply understanding how the bacterium could cause disease in a patient. The idea was to identify how the bacterium could evade detection by the immune system so that he could find a way to reveal it and prevent its sneak attacks.
It turns out, the bacterium can create a sort of cloaking device, which Geisinger calls a “capsule” or an “envelope.” That capsule is a sort of protective layer that allows the microbe to avoid being killed by the immune system.
Furthermore, Geisinger’s research has revealed that when the bacterium is exposed to antibiotics, it increases the production of this capsule layer to fortify the protective envelope. He also found that the wrong antibiotics can also make the bacterium shift from being fairly tame to quite aggressive in its ability to cause disease in a mouse, as well as even more resistant to antibiotic treatment.
So what doesn’t kill A. baumannii does indeed make it stronger.

There are two proteins that Geisinger found work together to regulate the bacterium’s ability to fortify the protective envelope against the immune system, resist antibiotic treatment, and increase its ability to cause disease. Those two proteins are now where Geisinger has turned his attention, supported by a research grant his laboratory was just awarded from the National Institutes of Health.
“The organism is really dependent on these two proteins,” he says. “This is how the organism protects itself.”
Geisinger’s goal is to find a way to inhibit the role of the pair of proteins to either develop new drugs or figure out how to make current antibiotics more potent against A. baumannii.
“If we could target one of the proteins, we could kill the whole system and make the organism more vulnerable,” he says. “If we can remove that layer of defense, we could hopefully expand the arsenal of treatments against this bug.”
For media inquiries, please contact Marirose Sartoretto at m.sartoretto@northeastern.edu or 617-373-5718.





